MAKE A MEME
View Large Image
| View Original: | P450cycle.svg (9239x6967) | |||
| Download: | Original | Medium | Small | Thumb |
| Courtesy of: | commons.wikimedia.org | More Like This | ||
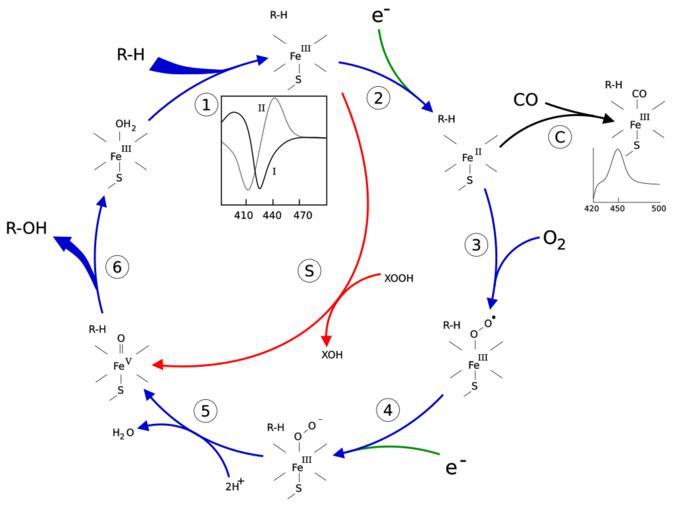
| Keywords: P450cycle.svg See https //en wikipedia org/wiki/User_talk Slashme/Archive_6 P450_Mechanism - not completely useless but should be used with care until it's fixed en The P450 catalytic cycle 1 The substrate binds to the active site of the enzyme in close proximity to the heme group on the side opposite to the peptide chain The bound substrate induces a change in the conformation of the active site displacing a water molecule from the distal axial coordination position of the heme iron<ref name P450Mechanism> cite journal Bernard Meunier Samuël P de Visser and Sason Shaik Mechanism of Oxidation Reactions Catalyzed by Cytochrome P450 Enzymes Chemical Reviews 104 9 3947 - 3980 2004 </ref> changing the state of the heme iron from low-spin to high-spin<ref name HiResCam> cite journal Thomas L Poulos Barry C Finzel and Andrew J Howard High-resolution crystal structure of cytochrome P450<sub>cam</sub> Journal of Molecular Biology 195 3 687-700 1987 </ref> This gives rise to a change in the spectral properties of the enzyme with an increase in absorbance at 390~nm and a decrease at 420~nm This can be measured by difference spectrometry and is referred to as the type~I difference spectrum see inset graph in figure Some substrates cause an opposite change in spectral properties a reverse type~I spectrum by processes that are as yet unclear Inhibitors and certain substrates that bind directly to the heme iron give rise to the type~II difference spectrum with a maximum at 430~nm and a minimum at 390~nm see inset graph in figure If no reducing equivalents are available this complex remains stable allowing the degree of binding to be determined from absorbance measurements in vitro<ref name p450struc> cite book P R Ortiz de Montellano Ed Cytochrome P450 structure mechanism and biochemistry 2nd ed Plenum 1995 New York </ref> 2 The change in the electronic state of the active site favours the transfer of an electron from NAD P H<ref name P450pot> cite journal S G Sligar D L Cinti G G Gibson and J B Schenkman Spin state control of the hepatic cytochrome P450 redox potential Biochemical and Biophysical Research Communications 90 3 925-932 1979 </ref> This takes place via the electron transfer chain as described above reducing the ferric heme iron to the ferrous state 3 Molecular oxygen binds covalently to the distal axial coordination position of the heme iron The cysteine ligand is a better electron donor than histidine with the oxygen consequently being activated to a greater extent than in other heme proteins However this sometimes allows the bond to dissociate the so-called decoupling reaction releasing a reactive superoxide radical interrupting the catalytic cycle<ref name P450Mechanism /> 4 A second electron is transferred via the electron-transport system reducing the dioxygen adduct to a negatively charged peroxo group This is a short-lived intermediate state 5 The peroxo group formed in step 4 is rapidly protonated twice by local transfer from surrounding amino-acid side chains releasing one mole of water and forming a highly reactive iron V -oxo species<ref name P450Mechanism /> 6 Depending on the substrate and enzyme involved P450 enzymes can catalyse any of a wide variety of reactions A hypothetical hydroxylation is shown in this illustration After the product has been released from the active site the enzyme returns to its original state with a water molecule returning to occupy the distal coordination position of the iron nucleus S An alternative route for mono-oxygenation is via the peroxide shunt interaction with single-oxygen donors such as peroxides and hypochlorites can lead directly to the formation of the iron-oxo intermediate allowing the catalytic cycle to be completed without going through steps 3 4 and 5<ref name p450struc /> A hypothetical peroxide XOOH is shown in the diagram C If carbon monoxide CO binds to reduced P450 the catalytic cycle is interrupted This reaction yields the classic CO difference spectrum with a maximum at 450 nm <references /> M Sc Thesis David Richfield User Slashme Slashme wikipedia en When using this image in external works it may be cited as follows cite journal Medical gallery of David Richfield 2014 Richfield David 1 2 Wikiversity Journal of Medicine 10 15347/wjm/2014 009 2001-8762 https //en wikiversity org/wiki/Medical_gallery_of_David_Richfield_2014 2008-03 Oxidoreductases Xenobiotic metabolism Enzyme mechanisms Cytochrome P-450 enzyme system | ||||